It remains at that temperature during the time that it changes to its liquid form, a process called melting. 
If the pail of ice is taken into a warm kitchen, it gradually grows warmer until it reaches a temperature of 32 ☏ (0 ☌). 
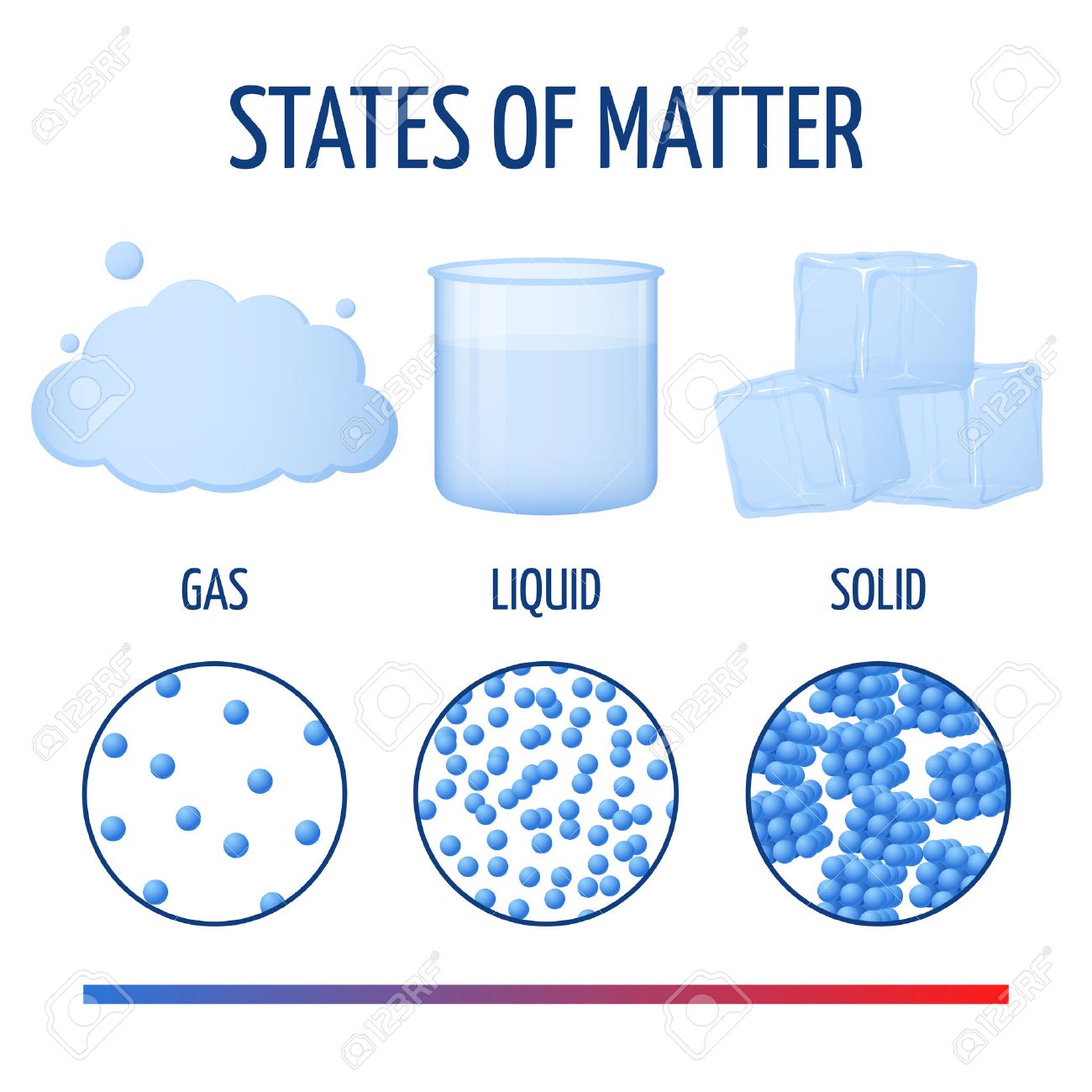
The state of a substance-that is, whether it is a solid, liquid, or gas-depends on its melting and freezing points. Individual grains of table salt are also solids at room temperature each keeps its own shape and size. However, water poured into a cup takes on the shape of the cup while it is in the cup.Īt room temperature, steel, copper, and diamonds are solids. Instead, the ice keeps its own size and shape. If a piece of ice is placed in a cup, it does not flow downward and take on the shape of the cup, as liquid water would do. It has changed from a liquid to a solid.Ī solid is a state of matter that maintains its own shape instead of conforming to the shape of its container. Its chemical structure-H 2O-remains the same, but its physical state is different. On freezing winter days, a pail of water left outdoors soon changes in the way it looks and feels. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
